We know that there are many projects and startups that are struggling and fighting for funding these days. We also believe that some projects deserve more urgent attention as they try to solve dramatic problems. For sure, the health of preterm babies must be a top priority. We are extremely happy to announce that our friends at Oxyprem have received an important financing support from the “SERI-funded EIC Accelerator Grant”.
Article and interview by Elsa María Granados Quirós
Globally, there are approximately 15 million premature live births every year (500,000 in Europe) and the numbers are expected to rise. 1 million out of these 15 million preterm babies die due to their prematurity and its consequences before they reach the age of five.
Because of their earlier birth, preterm babies have their respiratory and cardiovascular systems still underdeveloped and immature, which may affect the oxygen supply to their brains. Even slight variations of quantity in oxygen supply can cause irreversible damage such as neurodevelopmental impairment, paralysis and other disabilities, and even death.
Premature births are an increasingly worrisome issue. In addition to health consequences for babies, it Premature births are an increasingly worrisome issue. In addition to health consequences for babies, it has a big emotional and economic impact on their families and also has “implications for public-sector services, such as health insurance, educational, and other social support systems.” Preterm births can imply high lifetime costs, starting with the initial hospitalization costs, but also including educational costs and chronic disease treatment.
OxyPrem solution: the first preterm oximeter
Pre-term babies are treated with early oxygen (O2) therapy and, monitoring their brain oxygen level can help the clinical decision-making in order to avoid severe consequences. However, many devices which are commonly used for monitoring oxygen in preterm babies do not offer the precision that medical teams ask for. Moreover, they are often adapted devices initially designed for adults and thus they don’t meet the necessary characteristics for babies in this situation. The urgency to develop brain oximeters specifically designed for preterms has been extensively alerted by the world leading neonatologists.

This is where OxyPrem plays a crucial role. This Swiss company, based in Zurich, Switzerland, develops a medical device that ensures a high-precision measurement of the oxygen in premature babies’ brains. Thanks to its cutting-edge technology, OxyPrem’s device solves the difficulty of monitoring the oxygen supply in these delicate and challenging cases in which being aware of the smallest variations is of vital importance. “The oximeter consists of a near-infrared spectroscopy sensor embedded in biocompatible silicone”. This makes it flexible, adaptable and comfortable for the babies to be applied on their heads. It can be “fixed to the preterm’s head to monitor brain oxygen levels and to run analysis algorithms transforming the signals into tissue oxygenation saturation measurements”. According to CORDIS (Community Research and Development Information Service), “the oximeter prototypes have shown the highest levels of precision in clinical studies and are currently in the commercialisation phase.”
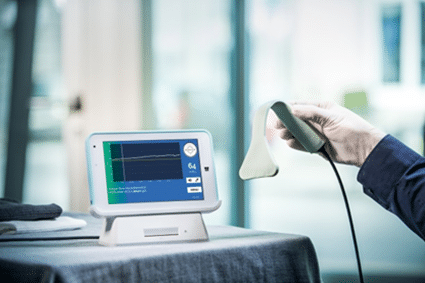
The project has undergone a long way and it continues its path to reach a larger market, aiming for European and North American hospitals. Here are some of the most important milestones of OxyPrem´s development, which have led the company to where it is now:
• Back in 2018, co-founder of OxyPrem, Alexander Nitsch, participated in the Academia-Industry Training (AIT) program of Swissnex India. Through this program, OxyPrem was coached and got further assistance, also connecting with key partners like St. John’s Medical College. This program made possible that OxyPrem oximeters got a first touch with India and are known to Indian specialist clinicians today.
• The connection with St. John’s Medical College led to a continuous engagement. Afterwards, OxyPrem and St. John’s partnered to participate in a breakthrough international research study: the SafeBoosC-III trial.
• In 2020, OxyPrem won the support of the EIC (European Innovation Council) Accelerator funding programme for the development of a feasibility analysis project. After accomplishing a 4-month feasibility study, OxyPrem was granted with a financial contribution of € 50,000. Strata helped OxyPrem obtain this first EIC grant and also contributed to the elaboration of the feasibility study, with a focus on the go-to-market strategy and funding roadmap.
• In 2021, OxyPrem got a GO at Step 2 in the EIC Accelerator program application. Nevertheless, OxyPrem was finally deemed ineligible, along with 23 other Swiss companies, due to unfruitful negotiations between the EU and the Swiss Government. The OxyPrem project and the other 23 were in the end,funded directly by the Swiss Government.
All these successful steps have been possible thanks to a unique technology and a great team and leaders : Dr. Fabienne di Gennaro (Quality and Regulatory Manager), Dr. Stefan Kleiser (CTO), Dr. Alexander Kalyanov (Product Engineer), Dr. Maximillian Murphy (Software Engineer), Alexander Nitsch (CEO), Anais Nüssli (Product Designer), Dr. Daniel Ostojic (Product Engineer & Risk Manager), Dr. Kristina Salzer (Product Manager), Margrit Teuscher Bernhard (Lead of Operations), Pieter van der Poel (Head of Marketing & Sales), David Vartok (Cloud Architect), Hanneke Verbiest (Post-Market Surveillance and Clinical Affairs Manager) and Prof. Martin Wolf (Chairman of the Board).
Strata has been working with OxyPrem for the last 3 years, supporting the company through the application process to the EIC Accelerator program. We continue working hand in hand with OxyPrem company in its next steps to help enhance its reach and we are very proud of being part of such a human and necessary project.
We have been talking with Alexander Nitsch, CEO of the OxyPrem’s team, to know a little more about the company’s journey and their experience working with Strata:
Could you tell us how everything began? And what motivated this journey?
With OxyPrem everything began more than 10 years ago when a very influential group of European neonatologists approached the University of Zurich. They asked for development of a device that can monitor the brain oxygen in preterm born children, and the reason was that, at the time, they were frustrated with the other equipment that was available to do that because it did not meet their clinical need. And, from them, the University of Zurich picked up the challenge and it went on for about ten years. The ending point of all that research was the foundation of OxyPrem as a company, because the University realized that the prototypes were so good that they should be brought into clinical practice. So they decided that there has to be a spin-off. And, this is, to this day, OxyPrem.
Presently, what stage (of commercialisation) is the project at?
We have CE marked equipment that is already in the market. We’ve used it within a big clinical study that ran over the last two years. We supplied many hospitals throughout Europe with our equipment, and our larger-scale commercialisation is planned to start in early 2023.
To what extent is the OxyPrem oximeter currently contributing to improve premature babies and their families’ lives? Could you give us recent numbers related to the impact made by OxyPrem?
There was a big clinical study that was running for two years to find out about the impact of brain oxygen monitoring on the outcome of the babies. There are no official figures there yet, but what I can tell you in terms of the numbers is that we supplied 28 hospitals in the whole of Europe and even one in India to participate in this big study. So, there is quite an impact because lots of clinicians adopted the product and said “we want to use this product for this study”.
Our device gives more information to the medical teams and, therefore, it also gives at least a feeling of comfort to the parents because they know that something is being done to help clinicians take good decisions, so that their children won’t get disabilities or might even die in the first days of their lives.
Has it been a challenging journey scientifically speaking? In your opinion, which technical and commercial milestones have been more challenging until today?
Scientifically speaking yes, it has been a challenging journey. You can just see it from the fact that it took over ten years of University research until the whole thing got to a point where it would be converted into a spin-off company.
From the physical principles there are behind the whole technology, it’s not very complicated. You just use light to look into the tissue and to find out about the oxygenation.
The difficulty in this one is just to make a design and to get a precision that actually makes a difference. Because, you have to be very careful with the patients and also in this fragile environment you have to make sure that you get a very, very good precision in the information that the sensor is collecting.
Commercial milestones I can’t really tell you anything about because commercialisation starts next year.
Would you like to share some special fact or anecdote about anything concerning OxyPrem?
A very encouraging thing that happened earlier this year was when we were physically visiting one of the key opinion leaders in the neonatal field. We showed them our sensor design, and we also told them about some plans that we have for the future. Hearing this, they said: “ok, if you can make this happen as you are telling now, I am ready to order five of your systems right now on the spot.” So, that was really encouraging to see how the medical teams are really wishing for a good solution in this field, because they really care about their patients.
What would you highlight about your collaboration with Strata?
I think that the overall vibe within the collaboration has always been really good. I mean the whole Strata team. We’ve always been in touch with David, we’ve been in touch with other several team members over the years, and the whole vibe was a very positive one. We’ve always had the impression that the Strata team is convinced that they can make it happen and, in the end, it worked.
Please, feel free to share anything you would like to, seizing the opportunity. It’s been a good experience. Last year, I recommended Strata to friends of mine who run another start-up.










